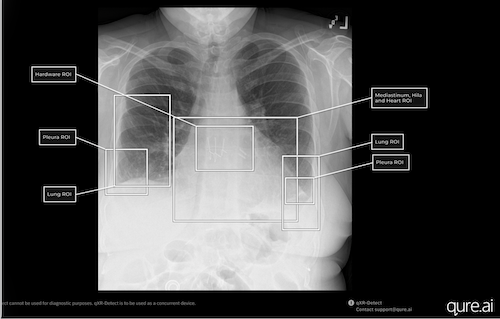
On February 26, 2026, the U.S. Food and Drug Administration (FDA) granted 510(k) class II clearance for Qure.ai’s qXR-Detect, a cutting-edge computer-assisted detection (CADe) solution for chest radiography. This technology aids in the early identification and categorization of significant findings on chest X-rays, benefiting emergency room physicians, family practitioners, and radiologists. With 70 million chest X-rays performed annually in the U.S., the FDA approval expands Qure.ai’s AI portfolio across six key regions of interest, enhancing the detection of over 100 potential abnormalities. Professor Amit Gupta emphasizes the importance of AI in visual localization, enabling radiologists to quickly assess alerts. Additionally, qXR-Detect is notable for its Predetermined Change Control Plan (PCCP), ensuring continuous updates. This latest clearance reflects Qure.ai’s commitment to innovation and regulatory excellence, solidifying its position in the AI medical imaging landscape. For more details, visit www.qure.ai.
Source link