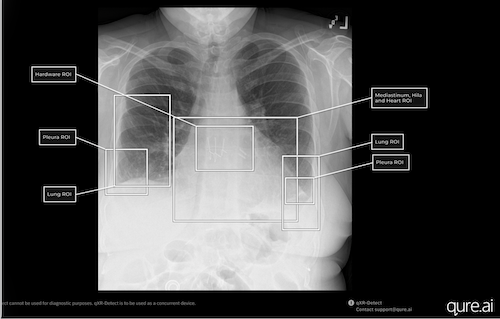
On February 26, 2026, the FDA granted 510(k) class II clearance for qXR-Detect, an advanced computer-assisted detection (CADe) solution by Qure.ai. Designed to enhance chest radiography, qXR-Detect assists emergency room physicians, family practitioners, and radiologists by facilitating the early identification and categorization of critical findings on chest X-rays, the most frequently performed imaging exams in the U.S. (70 million annually). This cutting-edge tool covers six key regions of interest, enabling the detection of over 100 potential abnormalities. Professor Amit Gupta cites the system’s visual localization features, which aid radiologists in quickly understanding alerts. Qure.ai’s technology, which also incorporates a Predetermined Change Control Plan (PCCP), ensures healthcare providers always utilize the latest versions of the algorithm. With 26 total FDA clearances across nine products, Qure.ai continues to lead in the AI radiology sector, supporting the pursuit of earlier lung cancer detection. For more information, visit www.qure.ai.
Source link